 :max_bytes(150000):strip_icc()/Periodic-Table-Metals-56a12db33df78cf772682c44.png)
“Effective nuclear charge” is a term used to describe the reduced charge felt by an electron due to shielding. Going down a group, although the number of protons increases, the number of core electrons also increases.Īt the end of each row (at the noble gases), the outermost shell has been filled and a new outermost shell begins at the start of the next row.Įlectron repulsion starts to become significant as electrons in lower shells push away electrons in higher shells.ĭue to the electron repulsion, the attractive forces of the nucleus is reduced. As the atomic number increases, the atomic radius also increases. Going down the group, a different trend unfolds. The atomic radius of atoms generally decreases More nuclear charge means the electrons are pulled closer to the nucleus, and hence reducing the atomic radius.The higher the atomic number, the higher the nuclear charge.Across each row, the atomic number increases but the energy level does not change (electrons are all added to the same shell).An atom with a higher nuclear charge and lower number of shells tends to attract the valence electrons closer to itself. The size of an atom is dictated by how far the valence electrons are from the nucleus of the atom. The major trends on the periodic table include: 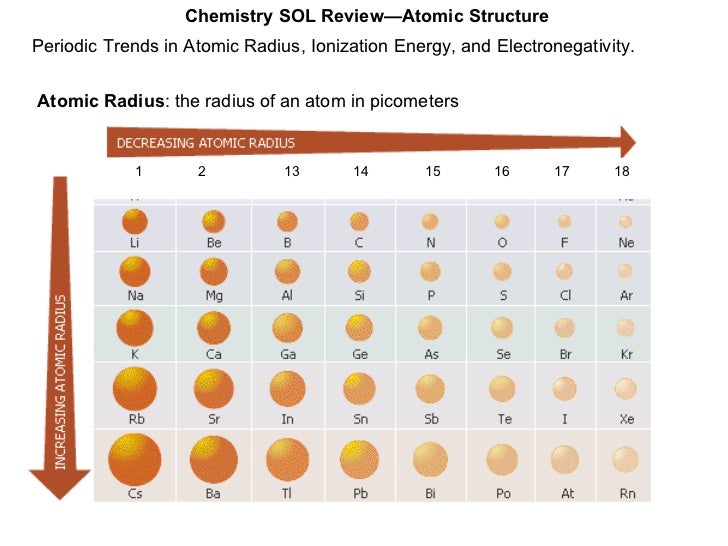
We will learn more about electronegativity in the electronegativity chapter. These electrons are called bonding electrons.ĭepending on which atom has a stronger pull, the pair of bonding electrons will move closer to that atom.Įlectronegativity is a measure of an atom’s ability to attract a pair of bonding electrons. When two atoms form a bond, a pair of electrons is involved. When chemical reactions take place, atoms play tug of war with the valence electrons. Valence electrons are those electrons found in the outermost shell of an atom. Only valence electrons can be bonding electrons. Any electron that’s not a valence electron.īonding electrons are electrons involved in chemical bonding. The inner electrons are called core electrons. Valence electrons are electrons in the highest energy level and are furthest away from the nucleus The nuclear charge is equal to the number of protons in the nucleus of an atom. Terminologiesīefore we delve into the periodic trends, let’s familiarize ourselves with a few new terms. This pattern is known as the periodic trends. Scientists discovered that if they arranged the elements according to their atomic number, properties of the elements would occur in a regular and repeating pattern. Today, we are going to learn about how the periodic table is organized and the important information that it provides. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
